Articles & Regulatory News in the Healthcare Sector
Read our articles, tips and updates from our world.
Parallel import of drugs into Switzerland: what does the law say?
Parallel importation of medicines is an issue of great interest to pharmaceutical…
Technical manager in Switzerland: requirements and responsibilities
In the Swiss regulatory environment for medical devices and regulated products, the…
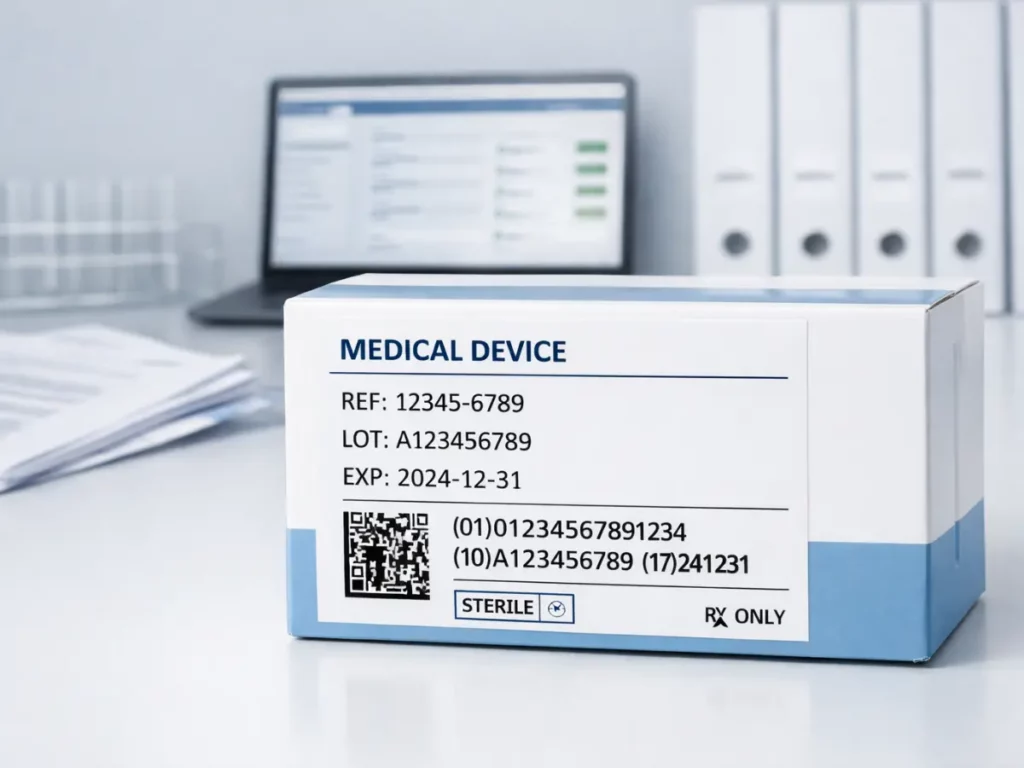
Traceability of Medical Devices: What is UDI and how it is used in Switzerland
Medical device traceability is now one of the central elements of regulatory…
GDP (Good Distribution Practice) in Switzerland: what it is, to whom it applies and how to implement it properly
In the life sciences sector, quality does not end with the production…
Nutraceuticals in Switzerland: rules, labeling and market opportunities
The market for nutraceuticals in Switzerland is growing steadily, driven by consumers…
How to effectively prepare for quality audits
A practical approach for pharmacies and life science companies Quality audits are…